Flotator Oxal as the plasticizer for suspension PVC Scientific paper
Main Article Content
Abstract
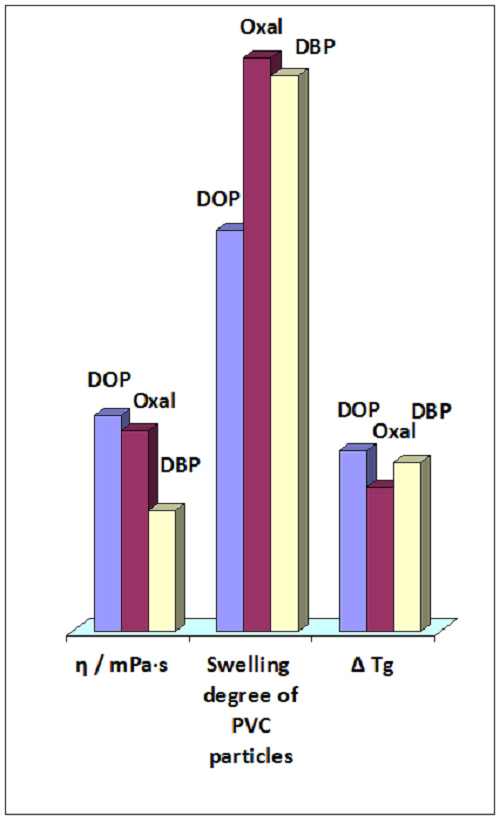
The flotator Oxal, mixture of dioxane ethers and alcohols as well as 1, 2 and 3 atomic alcohols, has been studied as a plasticizer for suspension PVC in comparison with the well-known dibutyl phthalate (DBP) and di-(2-ethylhexyl) phthalate (DOP). The rheological parameters and gelation ability of plasticizers were determined, the values of the storage modulus and tangent of mechanical loss angle in the glassy and rubbery states were measured by the DMA method, and the glass transition temperatures were determined. The deformation-strength properties and rigidity of polymer films were tested before and after light-thermal aging. Oxal was shown to reveal a fairly low viscosity and high gelation properties in relation to PVC. At the same time, its ability to reduce the glass transition temperature and elasticize the polymer in the glassy and rubbery state is somewhat lower than that of phthalate plasticizers. PVC samples plasticized with DBP have the highest resistance to light-thermal aging.
Downloads
Metrics
Article Details

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.

Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution license 4.0 that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal.
References
M. Rahman, C. S. Brazel, Prog. Polym. Sci. 29 (2004) 1223 (https://doi.org/10.1016/j.progpolymsci.2004.10.001)
K. Thinius, Chemie, Physik und Technologie der Weichmacher, 2nd ed., VEB Dt. Verl. für Grundstoffindustrie, Leipzig, 1963, p. 896
C. E. Wilkes, J. W. Summers, C. A. Daniels, M. T. Berard, PVC Handbook, 1st ed., Hanser Publications, Cincinnati, OH, 2005, p. 723 (ISBN 3-446-22714-8)
R. Navarro, M. P. Perrino, M. G. Tardajos, H. Reinecke, Macromolecules 43 (2010) 2377 (https://doi.org/10.1021/ma902740t)
V. Najafi, H. Abdollahi, Eur. Polym. J. 128 (2020) 109620 (https://doi.org/10.1016/j.eurpolymj.2020.109620)
V. V. Antic, M. N. Govedarica, J. Djonlagic, Polym. Int. 52 (2003) 1188 (https://doi.org/10.1002/pi.1241)
D. F. Cadogan, C. J. Howick, in: Ullmann's Encyclopedia of Industrial Chemistry, B. Elvers, Ed., Wiley-VCH, Weinheim, 2012, pp. 599–618 ( ISBN: 978-3-527-32943-4)
A. D. Godwin, in Applied Polymer Science: 21st Century, C. D. Craver, C. E. Carraher, Jr., Eds., Elsevier, New York, 2000, pp. 157–175 (ISBN 100080434177)
Plastics Additives Handbook, 5th ed., Hanser Gardner Publications, Cincinnati, OH, 2001, p. 1148 (ISBN-10: 1-56990295-X)
M. T. Benaniba, V. Massardier-Nageotte, J. Appl. Polym. Sci. 118 (2010) 3499 (https://doi.org/10.1002/app.32713)
M. Park, I. Choi, S. Lee, S. Hong, A. Kim, S. Jihoon, H.-C. Kang, Y.-W. Kim, J. Ind. Eng. Chem. 88 (2020) 148 (https://doi.org/10.1016/j.jiec.2020.04.007)
H. B. Pyeon, J. E. Park, D. H. Suh, Polymer Testing 63 (2017) 375 (https://doi.org/10.1016/j.polymertesting.2017.08.029)
B. Y. Yu, A. R. Lee, S.-Y. Kwak, Eur. Polym. J. 48 (2012) 885 (https://doi.org/10.1016/j.eurpolymj.2012.02.008)
I. Kostić, T. Anđelković, D. Anđelković, T. Cvetković, D. Pavlović, J. Serb. Chem. Soc. 83(2018) 1157 (https://doi.org/10.2298/JSC180423058K)
P. Jia, L. Hu, M. Zhang, G. Feng, Y. Zhou, Eur. Polym. J. 87 (2017) 209 (http://dx.doi.org/10.1016/j.eurpolymj.2016.12.023)
I. L. Glazko, O. P. Gur’yanova, S. V. Levanova, S. A. Kozlova, N. S. Neiman, Russ. J. Appl. Chem. 78 (2005) 972
A. A. Gudkov, E. M. Gotlib, T. Z. Lygina, Izv. Vyssh. Uchebn. Zaved. Khim. Khim. Tekhnol. 47 (2004) 104
E. M. Gotlib, R. V. Kozhevnikov, E. S. Ilyicheva, A. G. Sokolova, Bull. Kazan Technol. Univ. 4 (2013) 151
E. M. Gotlib, A. G. Sokolova, Composite materials, plasticized with EDOS, 1st ed., Paleotype, Moscow, 2012, p. 235
S. K. Ogorodnikov, G. S. Idlis, Isoprene production, Chemistry, Leningrad, 1973, p. 296
E. M. Gotlib, Waste and by-products of national economic production are raw materials for organic synthesis, Chemistry, Moscow, 1989, p. 212
A. Y. Malkin, A. I. Isayev, Rheology Concepts, Methods, and Applications, ChemTec Publishing, Toronto, 2012, p. 473 (https://doi.org/10.1016/C2011-0-04626-4)
Ya. Yu. Frenkel, The Kinetic Theory of Liquids Nauka, Moscow, 1975, p. 424
M. Y. Dolomatov, G. I. Nizamova, N. A. Zhuravleva, J. Eng. Phys. Thermophys. 90 (2017) 1020 (https://doi.org/10.1007/s10891-017-1652-4).