Ligands containing 7-azaindole functionality for inner-sphere hydrogen bonding: Structural and photophysical investigations Scientific paper
Main Article Content
Abstract
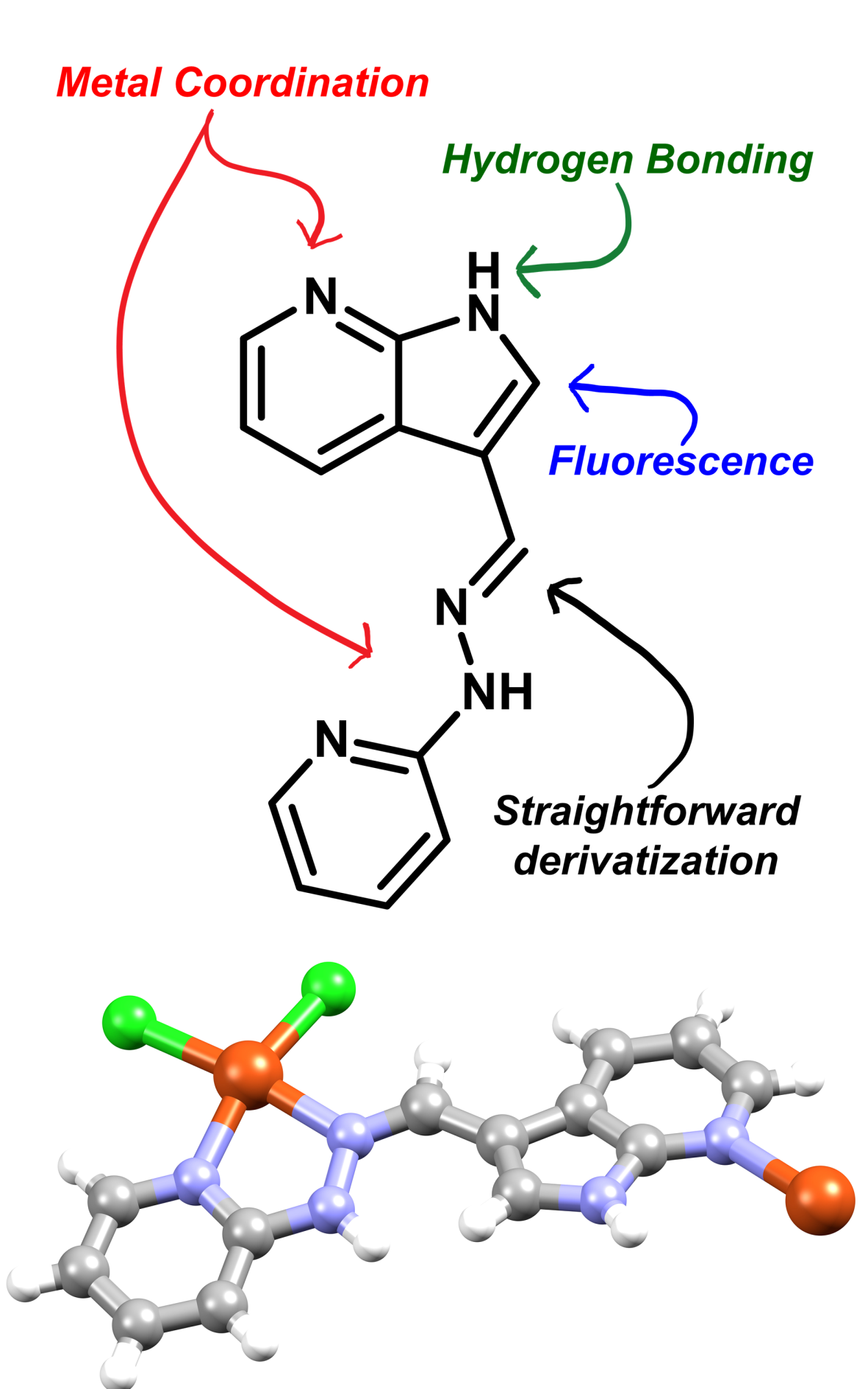
The synthesis, structural analysis and spectroscopic characterisation of three new 7-azaindole ligands is reported, alongside a novel 7-azaindole derived coordination polymer, with the aim of identifying new bridging ligands containing inner-sphere hydrogen bond donor functionality. Structural characterisation shows that the 7-azaindole hydrogen bond donor ability is significantly stronger in the hydrazone and imine species 1 and 2 compared to the amine 3, with the opposite trend evident in their hydrogen bond acceptor character. These findings are mirrored by the fluorescence spectroscopy results which show bimodal emission, characteristic of multiple emissive species related by proton transfer, is only evident in the amine species and not the more acidic imines. The polymeric copper(II) complex of the hydrazone ligand 1 shows the anticipated inner-sphere hydrogen bonding with a similar donor strength to that observed in the free ligand, which leads to deformation in the remainder of the coordination sphere. These results show the untapped versatility of the 7-azaindole functional group as a building block for ligands in coordination polymers and other multinuclear assemblies, with the potential for both stabilisation through hydrogen bonding and interesting photophysical properties.
Downloads
Metrics
Article Details

This work is licensed under a Creative Commons Attribution 4.0 International License.

Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution license 4.0 that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal.
Funding data
-
Royal Society of Chemistry
Grant numbers E21-4110373157 -
Keele University
Grant numbers School of Chemical and Physical Sciences
References
Z. Yin, Y.-L. Zhou, M.-H. Zeng, M. Kurmoo, Dalton Trans. 44 (2015) 5258 (https://doi.org/10.1039/C4DT04030A)
B. G. Diamond, L. I. Payne, C. H. Hendon, Commun. Chem. 6 (2023) 67 (https://doi.org/10.1038/s42004-023-00863-z)
M. Eddaoudi, J. Kim, N. Rosi, D. Vodak, J. Wachter, M. O’Keeffe, O. M. Yaghi, Science 295 (2002) 469 (https://doi.org/10.1126/science.1067208)
M. M. Radanović, S. B. Novaković, M. V. Rodić, L. S. Vojinović-Ješić, C. Janiak, V. M. Leovac, J. Serb. Chem. Soc. 87 (2022) 1259 (https://doi.org/10.2298/JSC220613072R)
P. D. Frischmann, V. Kunz, F. Würthner, Angew. Chem. Int. Ed. 54 (2015) 7285 (https://doi.org/10.1002/anie.201501670)
J. G. Betancourth, J. A. Castaño, R. Visbal, M. N. Chaur, Eur. J. Org. Chem. 2022 (2022) e202200228 (https://doi.org/10.1002/ejoc.202200228)
V. M. Leovac, V. S. Jevtović, L. S. Jovanović, G. A. Bogdanović, J. Serb. Chem. Soc. 70 (2005) 393 (https://doi.org/10.2298/JSC0503393L)
A. Erxleben, Inorg. Chim. Acta 472 (2018) 40 (https://doi.org/10.1016/j.ica.2017.06.060)
J.-F. Ayme, J. E. Beves, C. J. Campbell, D. A. Leigh, J. Am. Chem. Soc. 141 (2019) 3605 (https://doi.org/10.1021/jacs.8b12800)
S. Shaw, J. D. White, Chem. Rev. 119 (2019) 9381 (https://doi.org/10.1021/acs.chemrev.9b00074)
V. M. Leovac, M. V. Rodić, L. S. Jovanović, M. D. Joksović, T. Stanojković, M. Vujčić, D. Sladić, V. Marković, L. S. Vojinović-Ješić, Eur. J. Inorg. Chem. 2015 (2015) 882 (https://doi.org/10.1002/ejic.201403050)
A. Ferguson, M. A. Squire, D. Siretanu, D. Mitcov, C. Mathonière, R. Clérac, P. E. Kruger, Chem. Commun. 49 (2013) 1597 (https://doi.org/10.1039/C3CC00012E)
L. Fabbrizzi, J. Org. Chem. 85 (2020) 12212 (https://doi.org/10.1021/acs.joc.0c01420)
C. S. Hawes, Dalton Trans. 50 (2021) 6034 (https://doi.org/10.1039/D1DT00675D)
X.-L. Lv, S. Yuan, L.-H. Xie, H. F. Darke, Y. Chen, T. He, C. Dong, B. Wang, Y.-Z. Zhang, J.-R. Li, H.-C. Zhou, J. Am. Chem. Soc. 141 (2019) 10283 (https://doi.org/10.1021/jacs.9b02947)
K. Wang, Q. Wang, X. Wang, M. Wang, Q. Wang, H.-M. Shen, Y,-F. Yang, Y. She, Inorg. Chem. Front. 7 (2020) 3548 (https://doi.org/10.1039/D0QI00772B)
T. Basu, H. A. Sparkes, M. K. Bhunia, R. Mondal, Cryst. Growth Des. 9 (2009) 3488 (https://doi.org/10.1021/cg900195f)
C. S. Hawes, B. Moubaraki, K. S. Murray, P. E. Kruger, D. R. Turner, S. R. Batten, Cryst. Growth Des. 14 (2014) 5749 (https://doi.org/10.1021/cg501004u)
M. R. Healy, J. W. Roebuck, E. D. Doidge, L. C. Emeleus, P. J. Bailey, J. Campbell, A. J. Fischmann, J. B. Love, C. A. Morrison, T. Sassi, D. J. White, P. A. Tasker, Dalton Trans. 45 (2016) 3055 (https://doi.org/10.1039/C5DT04055H)
O. G. Wood, C. S. Hawes, CrystEngComm 24 (2022) 8197 (https://doi.org/10.1039/D2CE01475K)
R. S. Moog, M. Maroncelli, J. Phys. Chem. 95 (1991) 10359 (https://doi.org/10.1021/j100178a023)
X.-F. Yu, S. Yamazaki, T. Taketsugu, J. Chem. Theory Comput. 7 (2011) 1006 (https://doi.org/10.1021/ct200022a)
D. E. Folmer, E. S. Wisniewski, S. M. Hurley, A. W. Castleman Jr. Proc. Natl. Acad. Sci. U.S.A. 96 (1999) 12980 (https://doi.org/10.1073/pnas.96.23.12980)
A. V. Smirnov, D. S. English, R. L. Rich, J. Lane, L. Teyton, A. W. Schwabacher, S. Luo, R. W. Thornburg, J. W. Petrich, J. Phys. Chem., B 101 (1997) 2758 (https://doi.org/10.1021/jp9630232)
E. Wong, J. Li, C. Seward, S. Wang, Dalton Trans. (2009) 1776 (https://doi.org/10.1039/B814393E)
A. Domínguez-Martín, M. P. Brandi-Blanco, A. Matilla-Hernández, H. El Bakkali, V. M. Nurchi, J. M. González-Pérez, A. Castiñeiras, J. Niclós-Gutiérrez, Coord. Chem. Rev. 257 (2013) 2814 (https://doi.org/10.1016/j.ccr.2013.03.029)
B. Morzyk-Ociepa, K. Szmigiel, R. Petrus, I. Turowska-Tyrk, D. Michalska, J. Mol. Struct. 1144 (2017) 338 (https://doi.org/10.1016/j.molstruc.2017.05.059)
T. Steiner, Acta Crystallogr., B 57 (2001) 103 (https://doi.org/10.1107/S0108768100014348)
Y. Chen., R. L. Rich, F. Gai, J. W. Petrich, J. Phys. Chem. 97 (1993) 1770 (https://doi.org/10.1021/j100111a011)
P. Doungdee, S. Sarel, N. Wongvisetsirikul, S. Avramovici-Grisaru, J. Chem. Soc., Perkin Trans. 2 (1995) 319 (https://doi.org/10.1039/P29950000319)
M. C. Etter, J. C. MacDonald, J. Bernstein, Acta Crytallogr., B 46 (1990) 256 (https://doi.org/10.1107/S0108768189012929)
B. Morzyk-Ociepa, K. Dysz, I. Turowska-Tyrk, D. Michalska, J. Mol. Struct. 1101 (2015) 91 (https://doi.org/10.1016/j.molstruc.2015.08.003)
T. Beyer, S. L. Price, J. Phys. Chem., B 104 (2000) 2647 (https://doi.org/10.1021/jp9941413)
M. Nishio, CrystEngComm 6 (2004) 130 (https://doi.org/10.1039/B313104A)
H. Yokoyama, H. Watanabe, T. Omi, S. Ishiuchi, M. Fujii, J. Phys. Chem., A 105 (2001) 9366 (https://doi.org/10.1021/jp011245g)
H. Masui, S. Kanda, S. Fuse, Commun. Chem. 6 (2023) 47 (https://doi.org/10.1038/s42004-023-00837-1)
A. W. Addison, T. N. Rao, J. Reedijk, J. van Rijn, G. C. Verschoor, J. Chem. Soc., Dalton Trans. (1984) 1349 (https://doi.org/10.1039/DT9840001349)
G. Bartlett, I. Langmuir, J. Am. Chem. Soc. 43 (1921) 84 (https://doi.org/10.1021/ja01434a010)
C. A. Taylor, M. A. El-Bayoumi, M. Kasha, Proc. Natl. Acad. Sci. U.S.A. 63 (1969) 253 (https://doi.org/10.1073/pnas.63.2.253)
J. Konijnenberg, A. H. Huizer, C. A. G. O. Varma, J. Chem. Soc., Faraday Trans. 2 84 (1988) 1163 (https://doi.org/10.1039/F29888401163).