Investigation of adsorption properties of SF6 decomposed gases (SO2 and SO2F2) on pristine and Ti-decorated SWCNT surfaces: A DFT study Scientific paper
Main Article Content
Abstract
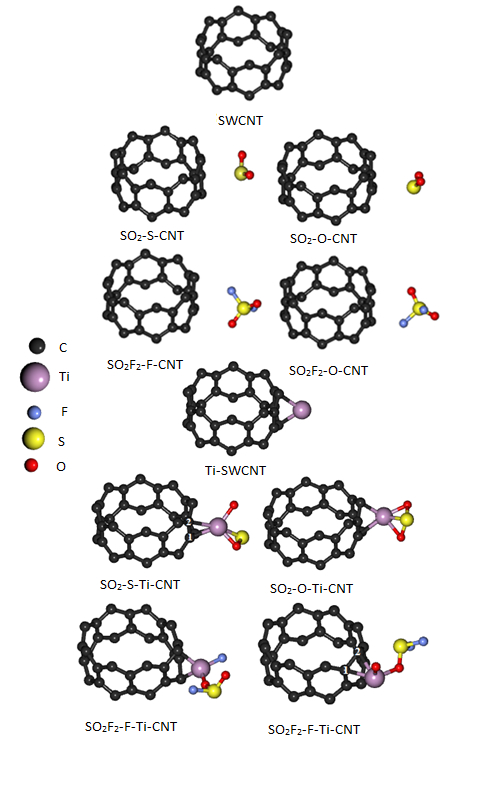
DFT calculations were employed to investigate the adsorption of gases produced from SF6 decomposition (SO2 and SO2F2) on pristine and Ti-decorated single-walled carbon nanotubes (Ti-(8,0) SWCNT). All structures were relaxed and their structural and electronic properties were investigated before and after gas adsorption on the surface of the nanotubes. (Ti-(8,0) SWCNT) was found to have high chemisorption sensitivity to Ti, SO2F2 and SO2 adsorptions on its surface. The electronic properties of (8,0) SWCNT were altered from semiconductor to metallic upon decoration with Ti, as demonstrated by the calculated band structures and the density of states (DOS). SO2F2 and SO2 adsorption on the surface of (Ti-(8,0) SWCNT) from different sides transformed the conductor (Ti-(8,0) SWCNT) into a semiconductor nanotube. To more carefully study the nature of adsorption, partial density of states (PDOS) calculations were also made. Additionally, Ti decoration induced a magnetization of approximately 2.61 μB in (8,0) SWCNT, which disappeared after gas adsorption.
DFT calculations were employed to investigate the adsorption of gases produced from SF6 decomposition (SO2 and SO2F2) on pristine and Ti-decorated single-walled carbon nanotubes (Ti-(8, 0) SWCNT). All structures were relaxed and their structural and electronic properties were investigated before and after gas adsorption on the surface of the nanotubes. (Ti-(8, 0) SWCNT) was found to have high chemisorption sensitivity to Ti, SO2F2, and SO2 adsorptions on its surface. The electronic properties of (8, 0) SWCNT were altered from semiconductor to metallic upon decoration with Ti, as demonstrated by calculated band structures and density of states (DOS). SO2F2 and SO2 adsorption on the surface of (Ti-(8, 0) SWCNT) from different sides transformed the conductor (Ti-(8, 0) SWCNT) into a semiconductor nanotube. To more carefully study the nature of adsorption, partial density of states (PDOS) calculations were also made. Additionally, Ti decoration induced a magnetization of approximately 2.61 μB in (8, 0) SWCNT, which disappeared after gas adsorption.
Downloads
Metrics
Article Details

This work is licensed under a Creative Commons Attribution 4.0 International License.

Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution license 4.0 that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal.
References
I. Sauers, H. W. Ellis, L. G. Christophorou, IEEE Trans. Elect. Insul. 2 (1986) 111 (https://doi.org/10.1109/TEI.1986.348932)
X. Zhang, B. Yang, W. Liu, J. Zhang, Proc. Eng. 29 (2012) 4107 (https://doi.org/10.1016/j.proeng.2012.01.628)
S. Iijima, Nature 354 (1991) 56 (https://doi.org/10.1038/354056a0)
D. R. Kauffman, A. Star, Angew. Chem. Int. Ed. 47 (2008) 6550 (https://doi.org/10.1002/anie.200704488)
R. Ströbel, J. Garche, P. T. Moseley, L. Jörissen, G. Wolf, J. Power Sources 159 (2006) 781 (https://doi.org/10.1016/j.jpowsour.2006.03.047)
S. Demir, M. F. Fellah, Surf. Sci. 701 (2020) 121689 (https://doi.org/10.1016/j.susc.2020.121689 )
J. Kong, N. R. Franklin, C. Zhou, M. G. Chapline, S. Peng, K. Cho, H. Dai, Science 287 (2000) 622 (https://doi.org/10.1126/science.287.5453.622)
J. Zhao, A. Buldum, J. Han, J. P. Lu, J. Nanotech. 13 (2002) 195 (https://doi.org/10.1088/0957-4484/13/2/312)
X. Zhou, W. Q. Tian, X. L. Wang, Sens. Actuators, B 151 (2010) 56 (https://doi.org/10.1016/j.snb.2010.09.054)
G. Gecim, & M. Ozekmekci, Surf. Sci. 711 (2021) 121876 (https://doi.org/10.1016/j.susc.2021.121876)
M. Patrignani, J. Juan, O. Nagel, W. Reimers, R. Luna, P. V. Jasen, Powr Tech. (2024) 119691 (https://doi.org/10.1016/j.powtec.2024.119691)
P. O. Krasnov, T. V. Basova, A. Hassan, Appl. Surf. Sci. 457 (2018) 235 (https://doi.org/10.1016/j.apsusc.2018.06.282)
W. Li, J. J. Ma, P. Liu, Z. L. Pan, Q. Y. He, Appl. Surf. Sci. 335 (2015) 17 (https://doi.org/10.1016/j.apsusc.2015.01.181)
L. L. Yu, S. Zhang, Q. Dong, X. Z. Meng, W. Q. Tian, J. Comput. Theor. Nanosci. 8 (2011) 1811 (https://doi.org/10.1166/jctn.2011.1887)
P. Giannozzi, S. Baroni, N. Bonini, M. Calandra, R. Car, C. Cavazzoni, D. Ceresoli, G. Chiarotti, M. Cococcioni, I. Dabo, A. DalCorso, S. deGironcoli, S. Fabris, G. Fratesi, R. Gebauer, U. Gerstmann, C. Gougoussis, A. Kokalj, M. Lazzeri, L. Martin-Samos, N. Marzari, F. Mauri, R. Mazzarello, S. Paolini, A. Pasquarello, L. Paulatto, C. Sbraccia, S. Scandolo, G. Sclauzero, A. Seitsonen, A. Smogunov, P. Umari, R. M. Wentzcovitch, J. Phys: Cond. Matter. 21 (2009) 395502 (https://doi.org/10.1088/0953-8984/21/39/395502)
J. P. Perdew, K. Burke, M. Ernzerhof, J. Phys. Rev. Lett. 77 (1996) 3865 (https://doi.org/10.1103/PhysRevLett.77.3865)
H. J. Monkhorst, J. D. Pack, J. Phys. Rev., B 13 (1976) 5188 (https://doi.org/10.1103/PhysRevB.13.5188)
S. F. Boys, F. G. M. P. Bernardi, J. Mol. Phys. 19 (1970) 553 (https://doi.org/10.1080/00268977000101561)
E. Sanville, S. D. Kenny, R. Smith, G. Henkelman, J. Comp. Chem. 28 (2007) 899 (http://doi.org/10.1002/jcc.20575)
W. Tang, E. Sanville, G. Henkelman, J. Phys.: Cond. Matter 21 (2009) 084204 (https://doi.org/10.1088/0953-8984/21/8/084204)
X. Zhang, Y. Gui, Z. Dai, Appl. Surf. Sci. 315 (2014) 196 (https://doi.org/10.1016/j.apsusc.2014.07.056)
X. Zhang, J. Zhang, J. Tang, B. Yang, J. Comp. Theor. Nanosci. 9 (2012) 1096 (https://doi.org/10.1166/jctn.2012.2149)
F. Mei, X. Ma, Y. Bie, G. Xu, J. Theor. Comp. Chem. 16 (2017) 1750065 (https://doi.org/10.1142/S0219633617500651)
K. W. Kayang, E. Nyankson, J. K. Efavi, V. K. Apalangya, B. I. Adetunji, G. Gebreyesus, A. Yaya, Res. Phys. 12 (2019) 2100 (https://doi.org/10.1016/j.rinp.2019.02.062)
X. Zhang, Y. Gui, H. Xiao, Y. Zhang, App. Surf. Sci. 379 (2016) 47 (https://doi.org/10.1016/j.apsusc.2016.04.048)
H. Huang, Y. Yu, M. Zhang, App. Surf. Sci. 505 (2020) 144622 (https://doi.org/10.1016/j.apsusc.2019.144622)
H. Cui, X. Zhang, D. Chen, J. Tang, App. Surf. Sci. 447 (2018) 594 (https://doi.org/10.1016/j.apsusc.2018.03.232).