Acetic acid liquid–liquid extraction and UHPLC-DAD detection of polycyclic aromatic hydrocarbons in toasted and fried foods Scientific paper
Main Article Content
Abstract
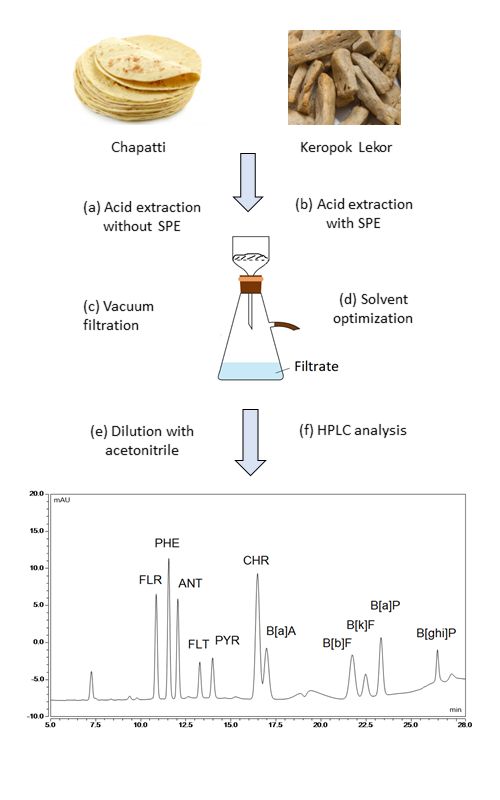
Polycyclic aromatic hydrocarbons (PAHs) are a large class of organic compounds containing two or more fused aromatic rings. They have very low water solubility and are highly lipophilic. Acetic acid is a polar protic solvent that is often used in reactions involving carbocation intermediates. In this study, acetic acid, coupled with other organic solvents, was optimized and used for the extraction of eleven PAHs in two types of foods: toasted and fried. The UHPLC-DAD method, coupled with a C18 column, was validated and applied to analyze eleven PAHs in the food samples. The LOD and LOQ values obtained ranged from 0.0049 to 0.373 μg L-1. The recoveries of the PAHs ranged from 47.3 to 119.7 %. The analysis results show that light PAHs were commonly found in both types of food. Some fried foods are highly carcinogenic due to the presence of BaP and Group 2B PAHs. Generally, toasted foods are safe to consume.
Downloads
Metrics
Article Details

This work is licensed under a Creative Commons Attribution 4.0 International License.

Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution license 4.0 that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal.
References
A. Onopiuk, K. Kołodziejczak, M. Marcinkowska-Lesiak, A. Poltorak, Food Chem. 373 (2022) 131506 (https://doi.org/10.1016/j.foodchem.2021.131506)
A. Zachara, D. Gałkowska, L. Juszczak, Int. J. Environ. Res. Public Health 15 (2017) 45 (https://doi.org/10.3390/ijerph15010045)
IARC Working Group on the Evaluation of Carcinogenic Risks to Humans, “IARC monographs on the evaluation of carcinogenic risks to humans. Some non-heterocyclic polycyclic aromatic hydrocarbons and some related exposures”, Vol. 92, IARC Monographs on Evaluation on Carcinogenic Risks to Humans, 2010, p. 773
European Communities, Amending Regulation (EC) no 1881/2006 as regards maximum levels for polycyclic aromatic hydrocarbons in food stuffs”, Commission Regulation (EC) no 835/2011
Commission Recommendation, (2005, February 4), “Further investigation into the levels of polycyclic aromatic hydrocarbons in certain foods”, Official Journal of the European Union.
L. K. Tintrop, A. Salemi, M. A. Jochmann, W. R. Engewald, T. C. Schmidt, Anal. Chim. Acta 1271 (2023) 341468 (https://doi.org/10.1016/j.aca.2023.341468)
H. V. Chuyen, M. H. Nguyen, P. D. Roach, J. B. Golding, S. E. Parks, Food Sci. Nutr. 6 (2018) 189 (https://doi.org/10.1002/fsn3.546)
C. S. Yue, A. K. Lim, M. L. Chia, P. Y. Wong, J. S. R. Chin, W. H. Wong, J. Food Sci. 88 (2023) 650 (https://doi.org/10.1111/1750-3841.16404)
V. Jalili, A. Barkhordari, A. Ghiasvand, Rev. Anal. Chem. 39 (2020) 1 (https://doi.org/10.1515/revac-2020-0101)
E. N. Hamidi, P. Hajeb, J. Selamat, S. Y. Lee, A. F. Abdull Razis, Int. J. Environ. Res. Public Health 19 (2022) 736 (https://doi.org/10.3390/ijerph19020736)
F. Cheng, B. Wang, Y. Xia, Int. J. Polym. Sci. 2018 (2018) 1 (https://doi.org/10.1155/2018/4960416)
T. O. A. Sturzoiu, T. Dobre, UPB Sci. Bull., B 68 (2006) 39
R. J. Ouellette, J. D. Rawn, Principles of Organic Chemistry, 1st ed., Elsevier, Amsterdam, 2015
P. Demmelmayer, L. Steiner, H. Weber, M. Kienberger, Sep. Purif. Technol. 310 (2023) 123060 (https://doi.org/10.1016/j.seppur.2022.123060)
M. J. Zarrinmehr, E. Daneshvar, S. Nigam, K. Panchamoorthy Gopinath, J. Kumar Biswas, E. E. Kwon, H. Wang, O. Farhadian, A. Bhatnagar, Bioresour. Technol. 344 (2022) 126303 (https://doi.org/10.1016/j.biortech.2021.126303)
J. O. Chaves, M. Corrêa de Souza, L. Capelasso da Silva, D. Lachos-Perez, P. C. Torres-Mayanga, A. P. da Fonseca Machado, T. Forster-Carneiro, M, Vázquez-Espinosa, A. Velasco González-de-Peredo, G. Fernández Barbero, M. Ariel Rostagno, Front. Chem. 8 (2020) 507887 (https://doi.org/10.3389/fchem.2020.507887)
M. Mahmoudpour, J. Mohtadinia, M.-M. Mousavi, M. Ansarin, M. Nemati, Food Anal. Methods 10 (2017) 277 (https://doi.org/10.1007/s12161-016-0579-2)
J. L. Navarro, M. Moiraghi, F. M. Quiroga, A. E. León, M. E. Steffolani, Food Technol. Biotechnol. 58 (2020) (https://doi.org/10.17113/ftb.58.03.20.6766)
H. Badibostan, J. Feizy, B. Daraei, S. Shoeibi, S. H. Rajabnejad, J. Asili, S. F. Taghizadeh, J. P. Giesy, G. Karimi, Food Chem. Toxicol. 131 (2019) 110640 (https://doi.org/10.1016/j.fct.2019.110640)
C. S. Yue, Q. N. Ng, A. K. Lim, M. H. Lam, K. N. Chee, Int. Food Res. J. 26 (2019) 999 (http://www.ifrj.upm.edu.my/26%20(03)%202019/30%20-%20IFRJ18644.R1-Final.pdf)
J. Cheng, J. Wang, F. Chen, D. Wu, C. Gao, W. Cheng, Z. Wang, X. Shen, X. Tang, Food Chem. 412 (2023) 135595 (https://doi.org/10.1016/j.foodchem.2023.135595)
C. P. Chong, H. Haron, S. Shahar, M. F. Md Noh, J. Food Compos. Anal. 80 (2019) 1 (https://doi.org/10.1016/j.jfca.2019.03.017).