Embracing green chromatography principles in perindopril, amlodipine and indapamide drug mixture analysis using β-cyclodextrin modified mobile phase
Main Article Content
Abstract
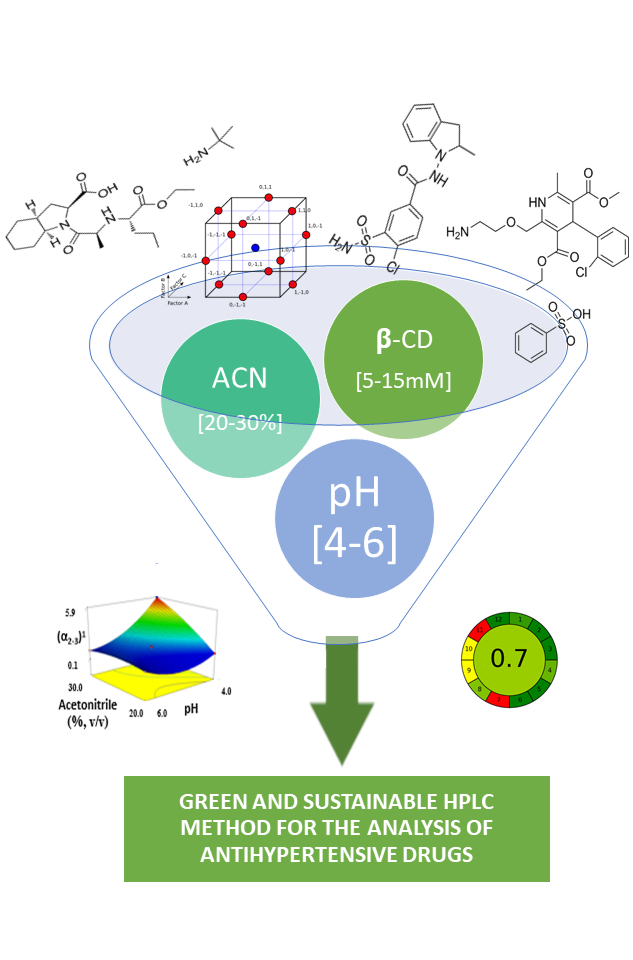
Raising the level of environmental awareness in the field of liquid chromatography is considered indispensable while the use of β-cyclodextrin in a mobile phase is promising strategy in this regard. This study presents a method development in lined with ICH Q14 regulatory requirements for introducing sustainability and method life-cycle management to separate components of a cardiovascular multi-drug tablet formulation. At the beginning, the analytical method target profile was defined, separation of perindopril, amlodipine, and indapamide in a shortest possible analytical run time. Following risk analysis pointed out that the mobile phase constituents represent the critical method parameters affecting the chromatographic analyses. Design of experiments methodology and Desirability function calculation was employed to simultaneously optimize the levels of concentration of β-cyclodextrin solution, pH value and acetonitrile content in the mobile phase investigated in the ranges 5-15 mM, 4.0-6.0 and 20-30% (v/v), respectively. The optimal chromatographic conditions consisted of 10 mM β-cyclodextrin (pH 5.4) and acetonitrile in the ratio 70:30 (v/v), 2 mL min-1 flow rate, RP-18e column kept at 25 °C, 215 nm detection wavelength, and 10 μL injection volume. The eco-friendliness of the method was assessed using the AGREE tool indicating a green and sustainable method was successfully developed.
Downloads
Metrics
Article Details

This work is licensed under a Creative Commons Attribution 4.0 International License.

Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution license 4.0 that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal.
Funding data
-
Ministarstvo Prosvete, Nauke i Tehnološkog Razvoja
Grant numbers 451-03-136/2025-03/200161;451-03-137/2025-03/200161
References
P. Anastas, Crit. Rew. Anal. Chem. 29 (2010) 167 (https://doi.org/10.1080/10408349891199356)
W. Löscher, Front. Pharmacol. 12 (2021) 730257 (https://doi.org/10.3389/fphar.2021.730257)
J. J. Lu, W. Pan, Y. J. Hu, Y. T. Wang, PLOS One 7 (2012) 40262 (https://doi.org/10.1371/journal.pone.0040262)
S. Julius, J. N. Cohn, J. Neutel, M. Weber, P. Turlapaty, Y. Shen, V. Dong, A. Batchelor, H. Lagast. J. Clin. Hypertens. 25 (2007) 10 (https://doi.org/10.1111/j.1524-6175.2004.02440.x)
T. Godfraind, J. Cardiovasc. Pharmacol. Ther. 19 (2014) 501 (https://doi.org/10.1177/1074248414530508)
D. B. Campbell, Curr. Med. Res. Opin. 8 (1983) 9 (https://doi.org/10.1185/03007998309109832)
United Nations General Assembly, Transforming our world: the 2030 Agenda for Sustainable Development, A/RES/70/1 (2015) (https://docs.un.org/en/A/res/70/1)
M. Yabré, L. Ferey, I. T. Somé, K. Gaudin, Molecules 23 (2018) 1065 (https://doi.org/10.3390/molecules23051065)
S. El Deeb, Molecules 29 (2024) 3205 (https://doi.org/10.3390/molecules29133205)
J. Stojanović, J. Krmar, B. Otašević, A. Protić, Arh. Farm. 73 (2023) 146 (https://doi.org/10.5937/arhfarm73-43479)
N. Đajić, J. Krmar, J. Stojanović, B. Svrkota, B. Otašević, A. Malenović, A. Protić, Arh Farm. 74 (2024) 235 (https://doi.org/10.5937/arhfarm74-49572)
F. Pena-Pereira, W. Wojnowski, M. Tobiszewski, Anal. Chem. 92 (2020) 10076 (https://doi.org/10.1021/acs.analchem.0c01887)
L. A. Yahya, C. Vakh, O. Dushna, O. Kalisz, S. Bocian, M. Tobiszewski, Anal. Chim. Acta 1357 (2025) 344052 (https://doi.org/10.1016/j.aca.2025.344052)
K. Patel, Sep. Sci. Plus 3 (2020) 175 (https://doi.org/10.1002/sscp.202000010)
K. N. Prashant, Y. Pragathi, S. Yenduri, Green Anal. Chem. 12 (2024) 100182 (https://doi.org/10.1016/j.greeac.2024.100182)
S. Agatonović-Kustrin, M. Lebete, M. E. Brown, D. W. Morton, B. Glass, J. Serb. Chem. Soc. 81 (2016) 1037 (https://doi.org/10.2298/JSC150915018A)
V. González-Ruiz, A. G. León, A. I. Olives, M. A. Martín, J. C. Menéndez, Green Chemistry 13 (2011) 115 (https://doi.org/10.1039/C0GC00456A)
N. Maljurić, J. Golubović, B. Otašević, M. Zečević, A. Protić, Anal. Bioanal. Chem. 410 (2018) 2533 (https://doi.org/10.1007/s00216-018-0911-3)
U. A. Thorsteinsdóttir, M. Thorsteinsdóttir, J. Mass Spectrom. 56 (2021) 4727 (https://doi.org/10.1002/jms.4727)
S. N. Hidayah, M. Mazidatu lakmam, S. M. F. Bukhari, R. N. Yani, J. Serb. Chem. Soc. 90 (2025) 1203 (https://doi.org/10.2298/JSC250124062S)
B. Svrkota, J. Krmar, F. Petronijević, A. Protić, B. Otašević, J. Sep. Sci. 48 (2025) 70136 (https://doi.org/10.1002/jssc.70136)
European medicinal agency, ICH Q14 Guideline on analytical procedure development (2023) (https://www.ema.europa.eu/en/ich-q14-analytical-procedure-development-scientific-guideline)
European medicinal agency, ICH Q2(R2) Guideline on validation of analytical procedures (2024) (https://www.ema.europa.eu/en/ich-q2r2-validation-analytical-procedures-scientific-guideline).