Extraordinary regularities of zinc dendrites’ growth under appropriate electrolysis conditions Scientific paper
Main Article Content
Abstract
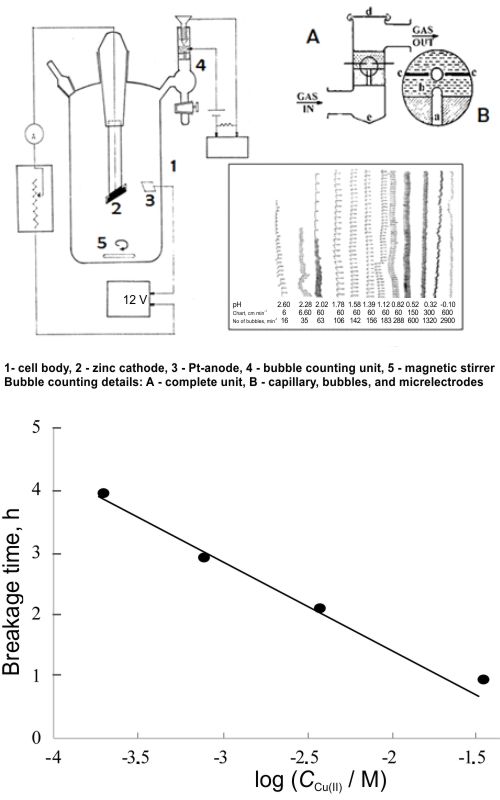
A case study is given of dendritic growth during zinc electrolysis in conditions that promote it. Subject of the main interest was how the electrolysis parameters affect the duration of dendrites life. The selected set of parameters did provide a surprisingly regular dendrite’s life, i.e., period from start of the electrolysis until dendrites detachment from the cathode. Dendrite’s growth did proceed with lowering of the zinc current efficiency, and the end of life was manifested by intensive hydrogen evolution due to corrosion of detached zinc deposit in the acid electrolyte. Current efficiency was successfully followed by the bubble counting technique, invented especially for kinetic studies of gas including reactions. The acquired results on dendrites’ life duration were so exact that it was easy to unify all five mono-variable dependencies into one five-variable expression. The calculated values of life duration did differ from the measured ones by only ±3 %! This is a proof that the developed expression accurately presents the real nature of dendritic growth under the applied conditions, i.e., 0.5 to 2 M zinc (II) ions, 0.41 to 3.06 M H2SO4, 10 to 2500 mg dm-3 copper (II) ions, 0.14 to 14 g dm-3 hexamethylenetetramine, and 4.25 to 103 mA dm-2 current density. The eventual broader region of Zn dendrites’ strict regular growth is not excluded.
Downloads
Metrics
Article Details

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.

Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution license 4.0 that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal.
References
V. A. Ettel, B. V. Tilak, in Electrochemical Processing, Comprehensive Treatise of Electrochemistry, Vol. 2, J. O’M. Bockris, B. E. Conway, E. Yeager, R. E. White, Eds., Plenum Press, New York, 1981, p. 78 (ISBN 978-1-4684-3787-4)
D. M. Dražić, S. Hadži Jordanov, Z. Nagy, Croat. Chem. Acta 45 (1973) 199 (https://hrcak.srce.hr/197061)
K. I. Popov, S. S. Djokić, B. N. Grgur, Fundamental aspects of electrometallurgy, Kluwer Academic Publishers, New York, 2002, pp. 72, 95 (ISBN 0-306-47269-4)
S. Zečević, S. Gojković, B. Nikolić, Electrochemical Engineering, Faculty of Technology and Metallurgy, Belgrade, 2001 (in Serbian)
M. M. Jakšić, Surf. Technol. 24 (1985) 193 (https://doi.org/10.1016/0376-4583(85)90071-8)
I. Ivanov, Hydrometallurgy 72 (2004) 73 (http://dx.doi.org/10.1016/S0304-386X(03)00129-4)
A. R. Despić, J. W. Diggle, J. O’M. Bockris, J. Electrochem. Soc. 115 (1968) 507 (https://doi.org/10.1149/1.2411297)
D. R. Hamilton, Electrochim. Acta 8 (1963) 731 (https://doi.org/10.1016/0013-4686(63)85018-5)
A. Milchev, Russ. J. Electrochem. 44 (2008) 619 (https://doi.org/10.1134/S1023193508060025)
E. A. Rosenberg, MSc. Thesis, NTNU, 2017 (http://hdl.handle.net/11250/2615762)
D. D. Gramatikov, MSc Thesis, Faculty of Technology and MetallurgySkopje, 1979, p. 92 (in Macedonian)
S. Hadži Jordanov, PhD Thesis, Faculty of Technology and Metallurgy, Belgrade, 1973 (in Serbian)
S. Hadži Jordanov, D. M. Dražić, Chem. Instrum. 6 (1975) 107
S. Hadži Jordanov, J. Naumovski, in Proceedings of Conference Corrosion and Cor¬rosion protection ’82, Central Council of STU in Bulgaria, Varna, Bulgaria, 1982, p. 32
S. Hadži Jordanov, in Proceedings of 7th International congress on metallic corrosion, Rio de Janeiro, Brazil, 1978, p. 365
A. C. Scott, R. M. Pitblado, G. W. Barton, in APCOM 87: Proceedings of the Twentieth International Symposium on the Application of Computers and Mathematics in the Min¬eral Industries, Vol. 2,South African Institute of Mining and Metallurgy in association with the Council for Mineral Technology, Johannesburg, South Africa, 1987, p. 51
S. Hadži Jordanov, P. Paunović, Electrolysis, Theory and Technology, Faculty of Technology and Metallurgy, Skopje, 2008 (in Macedonian).