Adsorption of tannase from Aspergillus ficuum to carboxyl-functionalized multi-walled carbon nanotubes Scientific paper
Main Article Content
Abstract
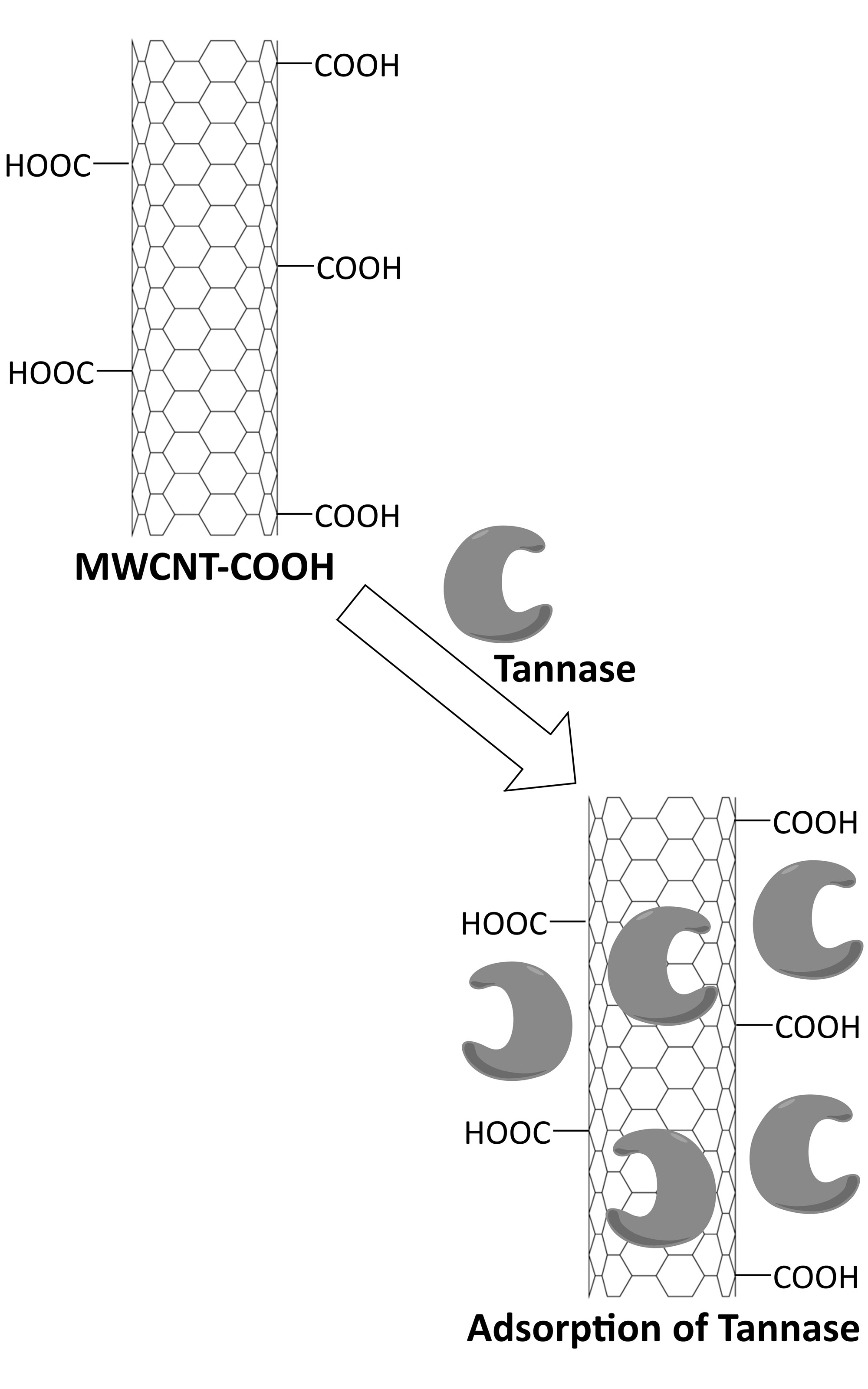
The immobilization of cross-linked tannase onto carboxyl-functionalized multi-walled carbon nanotubes (MWCNT-COOH) was achieved via physical adsorption. Glutaraldehyde was used to cross-link the enzyme molecules. Spectroscopic and morphological characterizations of the enzyme-nanotubes composite were carried out, which authenticated the successful adsorption event. Enzyme composite is proven equal to, or even superior than free tannase, in terms of catalytic activities and stabilities, when measured under different thermal, pH and recycling conditions. Whilst both free and immobilized tannase preparations exhibited optimum catalysis at pH 5.0 and 35 °C, tannase-nanotubes composite possesses better thermal stability. The immobilized preparation retained 75 % of its initial catalytic activity following ten consecutive uses. The study demonstrated a facile method to produce catalytically efficient nanobiocatalyst composite for biotechnological applications.
Downloads
Metrics
Article Details

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.

Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution license 4.0 that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal.
Funding data
-
Universiti Malaya
Grant numbers RMC/2021-22/03
References
A. S. Ristinmaa, T. Coleman, L. Cesar, A. Langborg Weinmann, S. Mazurkewich, G. Brändén, M. Hasani, J. Larsbrink, J. Biol. Chem. 298 (2022) (https://dx.doi.org/10.1016/j.jbc.2022.101758)
C. S. de Lima, B. T. A. Koelher, E. G. P. da Silva, A. Góes-Neto, R. P. Rezende, A. P. T. Uetanabaro, A. M. da Costa, Fungal Biology 126 (2022) 471 (https://dx.doi.org/10.1016/j.funbio.2022.04.001)
M. d. R. G. Dias, G. P. C. da Silva, A. de Pauloveloso, N. Krieger, C. Pilissão, Chirality 34 (2022) 1008 (https://dx.doi.org/10.1002/chir.23454)
M. Markiton, S. Boncel, D. Janas, A. Chrobok, ACS Sustain. Chem. Eng. 5 (2017) 1685 (https://dx.doi.org/10.1021/acssuschemeng.6b02433)
Z. L. Li, L. Cheng, L. W. Zhang, W. Liu, W. Q. Ma, L. Liu, Process Saf. Environ. Prot. 107 (2017) 463 (https://dx.doi.org/10.1016/j.psep.2017.02.021)
N. Singh, B. S. Dhanya, M. L. Verma, Mater. Sci. Energy Technol. 3 (2020) 808 (https://dx.doi.org/10.1016/j.mset.2020.09.006)
Y. Yamada, K. Obuchi, N. Kikuchi, A. A. Almarasy, A. Fujimori, Langmuir 38 (2022) 5692 (https://dx.doi.org/10.1021/acs.langmuir.2c00283)
A. Ulu, M. Karaman, F. Yapıcı, M. Naz, S. Sayın, E. İ. Saygılı, B. Ateş, Catal. Lett. 150 (2020) 1679 (https://dx.doi.org/10.1007/s10562-019-03069-y)
S. Zhao, P. Feng, Z. Yu, T. Zhou, T. Gao, M. M. Redina, P. Liu, X. Li, Chemosphere 291 (2022) 132934 (https://dx.doi.org/10.1016/j.chemosphere.2021.132934)
M. K. Ghide, K. Li, J. Wang, S. A. Abdulmalek, Y. Yan, Food Chem. 390 (2022) 133171 (https://dx.doi.org/10.1016/j.foodchem.2022.133171)
A. Singh, S. K. Rai, M. Manisha, S. K. Yadav, Mol. Catal. 511 (2021) 111723 (https://dx.doi.org/10.1016/j.mcat.2021.111723)
L. Y. Jun, N. M. Mubarak, L. S. Yon, C. H. Bing, M. Khalid, P. Jagadish, E. C. Abdullah, Sci. Rep. 9 (2019) 2215 (https://dx.doi.org/10.1038/s41598-019-39621-4)
R. Ahmad, S. K. Khare, Bioresour. Technol. 252 (2018) 72 (https://dx.doi.org/10.1016/j.biortech.2017.12.082)
S. Sharma, T. K. Bhat, R. K. Dawra, Anal. Biochem. 279 (2000) 85 (https://dx.doi.org/10.1006/abio.1999.4405)
M. Mohammadi, M. Ashjari, M. Garmroodi, M. Yousefi, A. A. Karkhane, RSC Advances 6 (2016) 72275 (https://dx.doi.org/10.1039/C6RA14142K)
C.-B. Ong, M. S. M. Annuar, Preparative Biochemistry & Biotechnology 48 (2018) 181 (https://dx.doi.org/10.1080/10826068.2018.1425707)
J. S. de Lima, M. P. Cabrera, C. M. de Souza Motta, A. Converti, L. B. Carvalho, Jr., Food Res. Int. 107 (2018) 470 (https://dx.doi.org/10.1016/j.foodres.2018.02.066)
R. Li, G. Fu, C. Liu, D. J. McClements, Y. Wan, S. Wang, T. Liu, Int. J. Biol. Macromol. 114 (2018) 1134 (https://dx.doi.org/j.ijbiomac.2018.03.077)
C. Wu, C. Xu, H. Ni, Q. Yang, H. Cai, A. Xiao, Bioresour. Technol. 205 (2016) 67 (https://dx.doi.org/10.1016/j.biortech.2016.01.032)
A. Xiao, C. Xu, Y. Lin, H. Ni, Y. Zhu, H. Cai, Electron. J. Biotechnol. 19 (2016) 1 (https://dx.doi.org/10.1016/j.ejbt.2015.10.001).