Oxidation of 1,5-benzodiazepine oximes catalysed by peroxidases Scientific paper
Main Article Content
Abstract
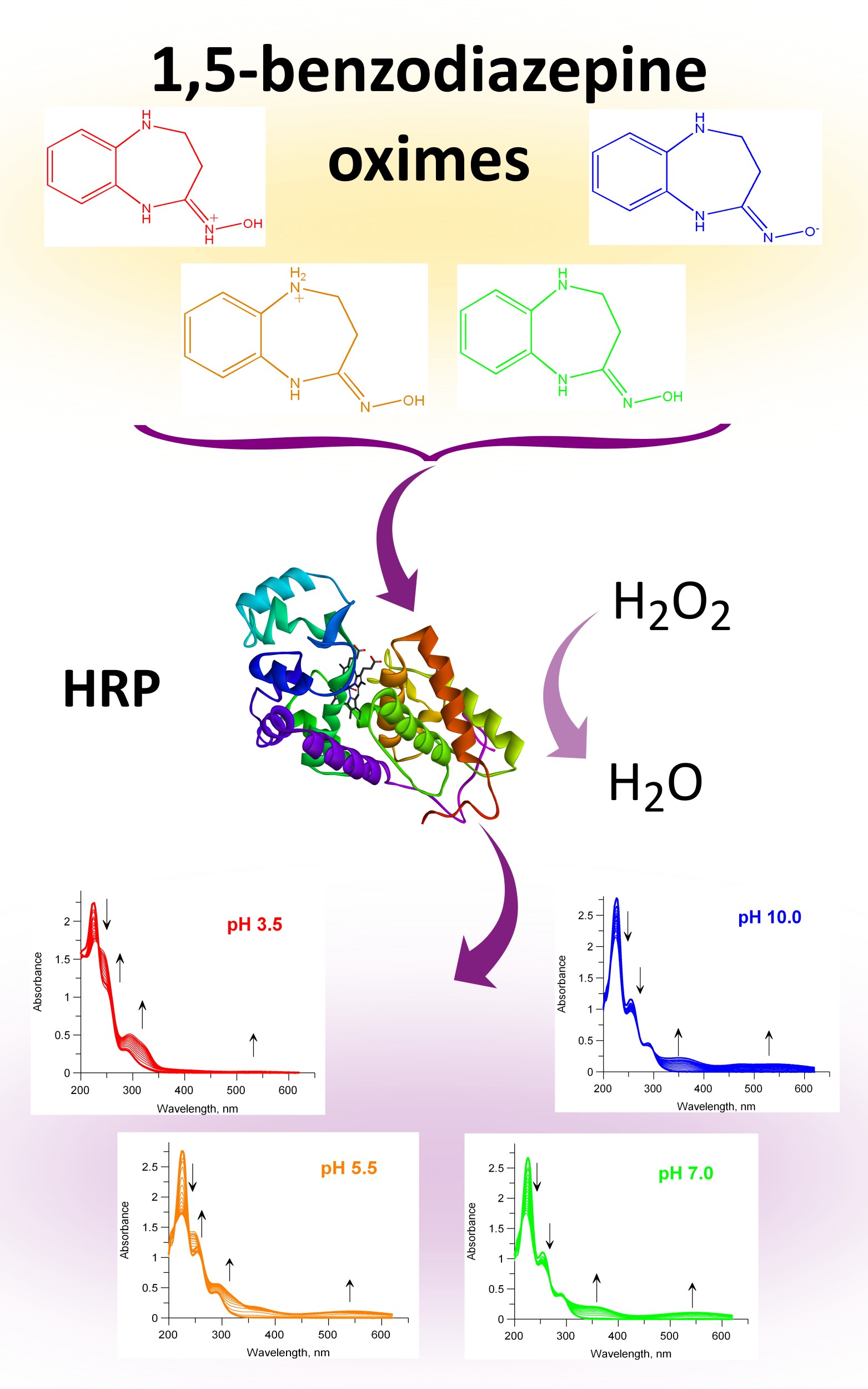
Oxidation of 1,3,4,5-tetrahydro-2H-1,5-benzodiazepine oximes catalysed by horseradish peroxidase (HRP) and recombinant Coprinus cinereus peroxidase (rCiP) was studied spectrophotometrically. The reaction rate dependences on the substrate and hydrogen peroxide concentrations were investigated; the values of apparent KM and Vmax, catalytic, oxidation and reduction constants (kcat, kox and kred, respectively) were calculated. The reactivity constants for the reactions catalysed by rCiP were higher than those for the HRP. Since oximes can have different structures depending on pH, the influence of pH on the rate of oxidation of compounds was studied. The dependences of the oxidation rate of the investigated oximes on the pH of the buffer solution were determined, and the pKa values of the amino acids of peroxidases responsible for the rate of catalysis were obtained. The HRP activity dependence on pH has a classical bell-shaped character, while rCiP dependence has a complex character.
Downloads
Metrics
Article Details

This work is licensed under a Creative Commons Attribution 4.0 International License.

Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution license 4.0 that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal.
References
W. B. Jakoby, Metabolic Basis of Detoxication: Metabolism of Functional Groups, Elsevier Science, Amsterdam, 1982 (ISBN: 9780323137997)
E. N. Shaw, 2-hydroxy-pyridine-N-oxide and process for preparing same, 1951 (http://www.google.com/patents/US2540218) Accessed May 8, 2023
N. K. F. W. Clauson-Kaas, N. Elming, J. Tormod Nielsen, Derivatives of N-hydroxy pyridines and process of production, 1956 (http://www.google.ch/patents/US2748142) Accessed May 8, 2023.
S. B. King, C-Nitroso Compounds, Oximes, N-Hydroxyguanidines and N-Hydroxyureas, John Wiley & Sons, Ltd., New York, 2005 (https://doi.org/10.1002/3527603751.CH7)
R. Rani, C. Granchi, Eur. J. Med. Chem. 97 (2015) 505 (https://doi.org/10.1016/J.EJMECH.2014.11.031)
P. Aastha, K. Navneet, A. Anshu, S. Pratima, K. Dharma, Res. J. Chem. Sci. 3 (2013) 90 (http://www.isca.me/rjcs/Archives/v3/i7/14.ISCA-RJCS-2013-057.php)
P. Jara-Ulloa, S. Catalán-Caro, C. A. Escobar, J. Chil. Chem. Soc. 59 (2014) 2520 (https://doi.org/10.4067/S0717-97072014000200027)
M. Whirl-Carrillo, R. Huddart, L. Gong, K. Sangkuhl, C. F. Thorn, R. Whaley, T. E. Klein, Clin. Pharmacol. Ther. 110 (2021) 563 (https://doi.org/10.1002/CPT.2350)
O. Mazimba, T. C. Molefe, Int. J. Chem. Stud. 3 (2015) 46 (https://www.chemijournal.com/archives/2015/vol3issue3/PartA/3-2-9.1.pdf)
L. Kosychova, R. Vidziunaite, G. Mikulskiene, I. Bratkovskaja, R. Janciene, Arkivoc 2015 (2015) 71 (https://doi.org/10.3998/ARK.5550190.P008.772)
S. Kumar, H. Kavitha, S. Arulmurugan, B. Venkatraman, Mini. Rev. Org. Chem. 9 (2012) 285 (https://doi.org/10.2174/1570193X11209030285)
K. D. Krewulak, H. J. Vogel, Biochim. Biophys. Acta – Biomembr. 1778 (2008) 1781 (https://doi.org/10.1016/J.BBAMEM.2007.07.026)
Y. Zhang, S. U. Geißen, C. Gal, Chemosphere 73 (2008) 1151 (https://doi.org/10.1016/j.chemosphere.2008.07.086)
T. Kosjek, S. Perko, M. Zupanc, M. Zanoški Hren, T. Landeka Dragičević, D. Žigon, B. Kompare, E. Heath, Water Res. 46 (2012) 355 (https://doi.org/10.1016/j.watres.2011.10.056)
T. Hata, H. Shintate, S. Kawai, H. Okamura, T. Nishida, J. Hazard. Mater. 181 (2010) 1175 (https://doi.org/10.1016/J.JHAZMAT.2010.05.103)
E. Marco-Urrea, M. Pérez-Trujillo, C. Cruz-Morató, G. Caminal, T. Vicent, Chemosphere 78 (2010) 474 (https://doi.org/10.1016/J.CHEMOSPHERE.2009.10.009)
A. Jelic, C. Cruz-Morató, E. Marco-Urrea, M. Sarrà, S. Perez, T. Vicent, M. Petrović, D. Barcelo, Water Res. 46 (2012) 955 (https://doi.org/10.1016/J.WATRES.2011.11.063)
S. Ostadhadi-Dehkordi, M. Tabatabaei-Sameni, H. Forootanfar, S. Kolahdouz, M. Ghazi-
-Khansari, M. A. Faramarzi, Bioresour. Technol. 125 (2012) 344 (https://doi.org/10.1016/J.BIORTECH.2012.09.039)
H. Kellner, P. Luis, M. J. Pecyna, F. Barbi, D. Kapturska, D. Krug̈er, D. R. Zak, R. Marmeisse, M. Vandenbol, M. Hofrichter, PLoS One 9 (2014) e95557 (https://doi.org/10.1371/JOURNAL.PONE.0095557)
K. G. Welinder, J. M. Mauro, & L. Norskov-Lauritsen, Biochem. Soc. Trans. 20 (1992) 337 (https://doi.org/https://doi.org/10.1042/bst0200337)
S.-J. Kim, J.-A. Lee, Y.-H. Kim, & B.-K. Song, J. Microbiol. Biotechnol. 19 (2009) 966 (https://doi.org/10.4014/jmb.0901.018)
A. K. Abelskov, A. T. Smith, C. B. Rasmussen, H. B. Dunford, K. G. Welinder, Biochemistry 36 (1997) 9453 (https://doi.org/10.1021/BI970387R)
Y. Yao, L. Huang, Y. Xu, Q. X. Li, J. Agric. Food Chem. 70 (2022) 646 (https://doi.org/10.1021/acs.jafc.1c06261)
R. Ivanec-Goranina, J. Kulys, Cent. Eur. J. Biol. 3 (2008) 224 (https://doi.org/10.2478/S11535-008-0021-X)
L. Rekovic, L. Kosychova, I. Bratkovskaja, R. Vidziunaite, J. Serb. Chem. Soc 83 (2018) 343 (https://doi.org/10.2298/JSC180226090R)
L. X. Shannon, E. Kay, J. Y. Lew, J. Biol. Chem. 241 (1966) 2166 (https://doi.org/https://doi.org/10.1016/S0021-9258(18)96680-9)
Z. S. Farhangrazi, I. Yamazaki, L. S. Powers, B. R. Copeland, T. Nakayama, T. Amachi, Biochemistry 33 (1994) 5647 (https://doi.org/https://doi.org/10.1021/bi00184a038)
D. P. Nelson, L. A. Kiesow, Anal. Biochem. 49 (1972) 474 (https://doi.org/https://doi.org/10.1016/0003-2697(72)90451-4)
E. Torres, M. Ayala, Biocatalysis based on heme peroxidases: Peroxidases as potential industrial biocatalysts, Springer, Berlin, 2010 (https://doi.org/https://doi.org/10.1007/978-3-642-12627-7_3)
B. M. Aveline, I. E. Kochevar, R. W. Redmond, J. Am. Chem. Soc. 118 (1996) 10124 (https://doi.org/https://doi.org/10.1021/ja961989l)
K. Fukunishi, K. Kitada, I. Naito, Synthesis (Stuttgart) 3 (1991) 237 (https://doi.org/https://doi.org/10.1055/s-1991-26433)
S. Sahu, S. Sahu, S. Patel, S. Dash, B. K. Mishra, Indian J. Chem. 47 (2008) 259. (https://nopr.niscpr.res.in/handle/123456789/1410)
M. Nissum, A. Feis, G. Smulevich, Biospectroscopy 4 (1998) 355 (https://doi.org/https://doi.org/10.1002/(SICI)1520-6343(1998)4:6<355::AID-BSPY1>3.0.CO;2-I)
P. Tn, T. L. Poulos, J. Kraut, J. Biol. Chem. 255 (1980) 8199 (https://doi.org/https://doi.org/10.1016/S0021-9258(19)70630-9)
G. Smulevich, A. Feis, C. Focardi, J. Tams, K. G. Welinder, Biochemistry 33 (1994) 15425 (https://doi.org/https://doi.org/10.1021/BI00255A024)
S. Ramalingam, M. Karabacak, S. Periandy, N. Puviarasan, D. Tanuja, Spectrochim. Acta, A 96 (2012) 207 (https://doi.org/10.1016/j.saa.2012.03.090)
S. Hashimoto, Y. Tatsuno, T. Kitagawa, Proc. Natl. Acad. Sci. U. S. A. 83 (1986) 2417 (https://doi.org/https://doi.org/10.1073/PNAS.83.8.2417)
I. Yamazaki, M. Tamura, & R. Nakajima, Mol. Cell. Biochem. 40 (1981) 143 (https://doi.org/https://doi.org/10.1007/BF00224608).