Synthesis of novel N-substituted benzyl N-(1,3-benzothiazol-2-yl) acetamides and their in vitro antibacterial activities Scientific paper
Main Article Content
Abstract
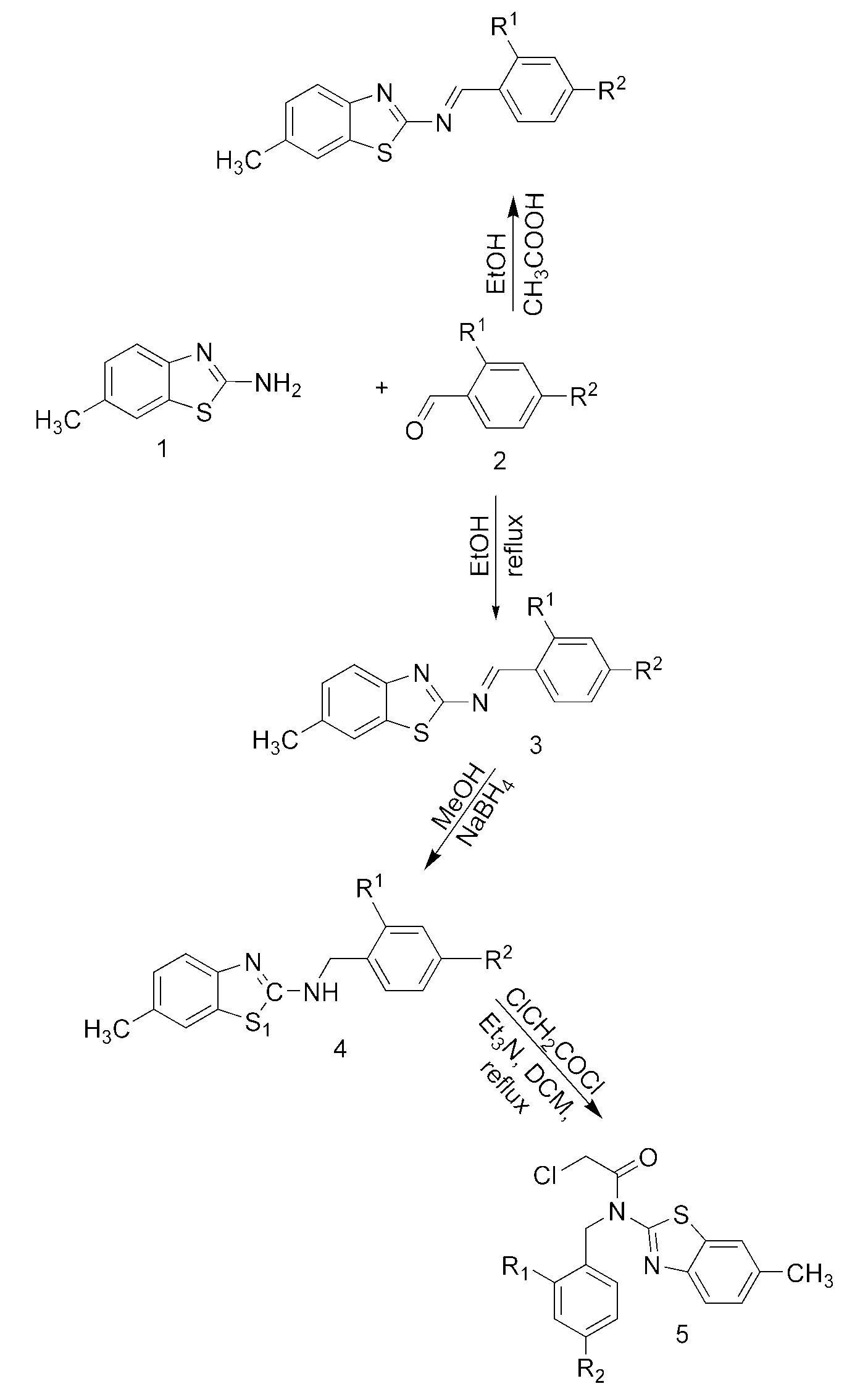
The novel Schiff bases 3a–d were synthesized by reacting 6-methyl-2-aminobenzothiazole and different substituted benzaldehydes. Afterwards, the obtained Schiff bases were reduced with NaBH4 to form amine compounds 4a–d. In the final step, reaction of the amine with chloroacetyl chloride gave the novel amide derivatives 5a–d. The structures of the all novel synthesized compounds were characterized by FT-IR, 1H-NMR, 13C-NMR, ESI MS, HETCOR, 2D (1H–1H) COSY spectra and elemental analyses. The antimicrobial activities of the novel synthesized compounds, were tested against some Gram-positive and Gram-negative bacterial as well as fungal species and the results were discussed.
Downloads
Metrics
Article Details

This work is licensed under a Creative Commons Attribution 4.0 International License.

Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution license 4.0 that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal.
Funding data
-
Eskişehir Osmangazi Üniversitesi
Grant numbers 200419032
References
H. F. Abd El-halim, M. M. Omar, G. G. Mohamed, Spectrochim. Acta, A 78 (2011) 36 (https://doi.org/10.1016/j.saa.2010.06.003)
T. B. S. A. Ravoof, K. A. Crouse, M. I. M. Tahir, F. N. F. How, R. Rosli, D. J. Watkins, Transit. Met. Chem. 35 (2010) 871 (https://doi.org/10.1007/s11243-010-9406-6)
A. Aragón-Muriel, Y. Liscano, Y. Upegui, S. M. Robledo, M. T. Ramírez-Apan, D. Morales-Morales, J. Oñate-Garzón, D. Polo-Cerón, Antibiotics 10 (2021) 728 (https://doi.org/10.3390/antibiotics10060728)
C. Ramalingan, S. Balasubramanian, S. Kabilan, M. Vasudevan, Eur. J. Med. Chem. 39 (2004) 527 (https://doi.org/10.1016/j.ejmech.2004.02.005)
G. T. Zitouni, S. Demirayak, A. Ozdemir, Z. A. Kaplancikli, M. T. Yildiz, Eur. J. Med. Chem. 39 (2004) 267 (https://doi.org/10.1016/j.ejmech.2003.11.001)
M. A. Neelakantan, S. S. Marriappan, J. Dharmaraja, T. Jeyakumar, K. Muthukumaran, Spectrochim. Acta, A 71 ( 2008) 628 (https://doi.org/10.1016/j.saa.2008.01.023)
M. T. Tarafder, A. Kasbollah, N. Saravan, K. A. Crouse, A. M. Ali, O. K. Tin, J. Biochem. Mol. Biol. Biophys. 6 (2002) 85 (https://doi.org/10.1080/10258140290027207)
I. Kucukguzel, S. G. Kucukguzel, S. Rollas, G. O. Sanis, O. Ozdemir, I. Bayrak, T. Altug, J. P. Stables, Farmaco 59 (2004) 893 (https://doi.org/10.1016/j.farmac.2004.07.005)
P. Vicini, A. Geronikaki, M. Incerti, B. Busonera, G. Poni, C. A. Cabras, P. La Colla, Bioorg. Med. Chem. 11 (2003) 4785 (https://doi.org/10.1016/S0968-0896(03)00493-0)
R. Pignatello, A. Panico, P. Mazzone, M. R. Pinizzotto, A. Garozzo, P. M. Fumeri, Eur. J. Med. Chem 29 (1994) 781 (https://doi.org/10.1016/0223-5234(94)90137-6)
S. Samadhiya, A. Halve, Orient. J. Chem. 17 (2001) 119
G. Mustafa, M. Zia-ur-Rehman, S. H. Sumrra, M. Ashfaq, W. Zafar, M. Ashfaq, J. Mol. Struct. 1262 (2022) 133044 (https://doi.org/10.1016/j.molstruc.2022.133044)
A. Ayati, S. Emami, A. Asadipour, A. Shafiee, A. Foroumadi, Eur. J. Med. Chem. 97 (2015) 699 (https://doi.org/10.1016/j.ejmech.2015.04.015)
a) F. Parlati, U. V. Ramesh, R. Singh, D. G. Payan, R. Lowe, G. C. Look, PCT Int. Appl. WO 2005037845, 2005p, b) M. Yoshida, I. Hayakawa, N. Hayashi, T. Agatsuma, Y. Oda, F. Tanzawa, S. Iwasaki, K. Koyama, H. Furukawa, S. Kurakata, Y. Sugano, Bioorg. Med. Chem. Lett. 15 (2005) 3328 (https://doi.org/10.1016/j.bmcl.2005.05.077) c) T. R. Bailey, D. C. Pevear, PCT Int. Appl. 2004078115, 2004p, d) A. Alanine, A. Flohr, A. K. Miller, R. D. Norcross, C. Riemer, PCT Int. Appl. WO 2001097786, 2001p; e) S. Kerwin, L. H. Hurley, M. R. De Luca, B. M. Moore, PCT Int. Appl. WO 9748694, 1997p; f) A. R. Brade, H. B. Khadse, A. S. Bobade, Indian Drugs 35 (1998) 554
D. Pareek, M. Chaudhary, P. K. Pareek, R. Kant, K. G. Ojha, R. Pareek, S. M. U. Iraqi, A. Pareek, Chem. Sin. 3 (2010) 22
A. Dandia, A. K. Jain, A. K. Laxkar, Tetrahedron Lett. 54 (2013) 3929 (https://doi.org/10.1016/j.tetlet.2013.05.035)
J. H. Billman, A. C. Diesing, J. Org. Chem. 22 (1957) 1068 (https://doi.org/10.1021/jo01360a019)
G. N. Walker, M. A. Klett, J. Med. Chem. 9 (1966) 624 (https://doi.org/10.1021/jm00322a049)
Y. Davion, G. Guillaumet, J.-M. Léger, C. Jarry, B. Lesur, J.-Y. Mérour, Heterocycles 63 (2004) 1093 (https://doi.org/10.3987/com-04-10022)
NCCLS, National Committee for Clinical Laboratory Standards, 2008, p. 120
F. Savaroglu, S. Ilhan, C. Filik-Iscen, J. Med. Plants Res. 5 (2011) 3286
F. Savaroglu, C. Filik Iscen, A.P. Oztopcu Vatan, S. Kabadere, S. Ilhan, R. Uyar, Turk. J. Biol. 35 (2011) 361 (https://doi.org/10.3906/biy-0906-46)
F. R. Deleo, B. A. Diep, M. Otto, Inf. Dis. Clin. N. Amer. 23 (2009) 17 (https://doi.org/10.1016/j.idc.2008.10.003)
A. Pietrzak, G. Chodorowska, I. Jazienicka, P. Osemlak, B. Wawrzycki, P. Terlecki, J. Mieczkowska, J. Mosiewicz, T. Zubilewicz, F. Szubstarski, W. Krupski, J. Hercogova, J. C. Szepietowski, T. Lotti, Derma. Ther. 24 (2011) 587 (https://doi.org/10.1111/j.1529-8019.2012.01410.x)
Y. Yang, E. J. Anderson, J. Appl. Microbiol. 86 (1999) 211 (https://doi.org/10.1046/j.1365-2672.1999.00652.x).