Synthesis and biological activity of novel zingerone–thiohydantoin hybrids Scientific paper
Main Article Content
Abstract
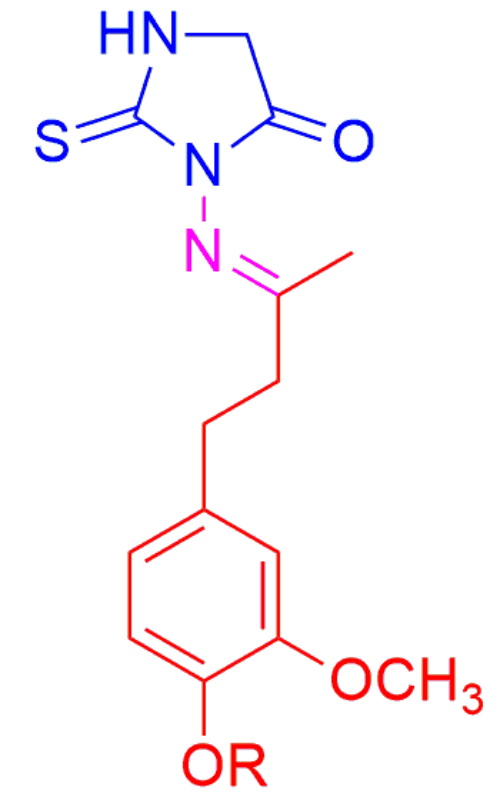
A series of zingerone–thiohydantoin hybrids were synthesized from O-alkyl zingerone derivatives by cyclocondensation with thiosemicarbazide in a two-step reaction. The obtained new potentially bioactive compounds were structurally characterized by IR and NMR spectroscopy, as well as by elemental and HRMS analysis. In addition, their antimicrobial and in vitro anticancer activities were tested. The tested compounds showed low to moderate antimicrobial activity. Zingerone–thiohydantoin hybrid with an O-butyl substituent exerted the significant cytotoxic activity on colon HCT-116 cancer cells, without toxicity on healthy MRC-5 cells.
Downloads
Metrics
Article Details

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.

Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution license 4.0 that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal.
Funding data
-
Ministarstvo Prosvete, Nauke i Tehnološkog Razvoja
Grant numbers 451-03-68/2022-14/200378;451-03-68/2022-14/200122
References
G. Bérubé, Expert Opin. Drug Discov. 11 (2016) 281 (https://www.doi.org/10.1517/17460441.2016.1135125)
V. A. S. Jesudoss, S. Victor Antony Santiago, K. Venkatachalam, P. Subramanian, in Gastrointestinal Tissue: Oxidative Stress and Dietary Antioxidants, J. Gracia-Sancho, J. Salvadó, Eds., Academic Press, Cambridge, MA, 2017, p. 289 (https://www.doi.org/10.1016/B978-0-12-805377-5.00021-7)
M. A. Metwally, E. Abdel-Latif, J. Sulfur Chem. 33 (2012) 229 (https://www.doi.org/10.1080/17415993.2011.643550)
P. G. C. de Carvalho, J. M. Ribeiro, R. P. B. Garbin, G. Nakazato, S. F. Yamada Ogatta, Â. de Fátima, M. de Lima Ferreira Bispo, F. Macedo, Lett. Drug Des. Discov. 17 (2020) 94 (https://www.doi.org/10.2174/1570180816666181212153011)
R. M. Gesler, C. E. Lints, E. A. Swinyard, Toxicol. Appl. Pharmacol. 3 (1961) 107 (https://doi.org/10.1016/0041-008X(61)90014-X)
L. Somsák, L. Kovács, M. Tóth, E. Ösz, L. Szilágyi, Z. Györgydeák, Z. Dinya, T. Docsa, B. Tóth, P. Gergely, J. Med. Chem. 44 (2001) 2843 (https://www.doi.org/10.1021/jm010892t)
S. Rajamaki, A. Innitzer, C. Falciani, C. Tintori, F. Christ, M. Witvrouw, Z. Debyser, S. Massa, M. Botta, Bioorganic Med. Chem. Lett. 19 (2009) 3615 (https://www.doi.org/10.1016/j.bmcl.2009.04.132)
M. Zuo, X. Xu, Z. Xie, R. Ge, Z. Zhang, Z. Li, J. Bian, Eur. J. Med. Chem. 125 (2017) 1002 (https://www.doi.org/10.1016/j.ejmech.2016.10.049)
L. R. Smith, Chem. Educ. 1 (1996) 1 (https://doi.org/10.1007/s00897960034a)
J. S. Buck, Org. Synth. 13 (1933) 102 (https://www.doi.org/10.15227/orgsyn.013.0102)
R. Katritzky, Q. Long, H. Y. He, G. Qiua, A. L. Wilcox, Arkivoc 2000 (2000) 868 (https://www.doi.org/10.3998/ark.5550190.0001.603)
Šmit, R. Z. Pavlović, A. Radosavljević-Mihailović, A. Došen, M. G. Ćurčić, D. S. Šeklić, M. N. Živanović, J. Serb. Chem. Soc.78 (2013) 217 (https://www.doi.org/10.2298/JSC120725154S)
S. D. Sarker, L. Nahar, Y. Kumarasamy, Methods 42 (2007) 321 (https://www.doi.org/10.1016/j.ymeth.2007.01.006)
Halilagić, E. Selimović, J. S. K. Stanković, N. Srećković, K. Virijević, M. N. Živanović, B. Šmit, T. V. Soldatović, J. Coord. Chem. (2022) 1 (https://www.doi.org/10.1080/00958972.2022.2048376)
K. Hostanska, G. Jürgenliemk, G. Abel, A. Nahrstedt, R. Saller, Cancer Detect. Prev. 31 (2007) 129 (https://www.doi.org/10.1016/j.cdp.2007.03.001)
T. Mosmann, J. Immunol. Methods 65 (1983) 55 (https://www.doi.org/10.1016/0022-1759(83)90303-4)
R. A. Mekheimer, A. M. A. Hameed, K. U. Sadek, Molecules 13 (2008) 195 (https://doi.org/10.3390/molecules13010195)
G. Sprengler, J. Handzlik, I. Ocsovszki, M. Viveiros, K. Kiec-Kononowicz, J. Molnar, L. Amaral, Anticancer Res. 31 (2011) 3285 (https://ar.iiarjournals.org/content/31/10/3285)
R. Vinothkumar, R. Vinothkumar, M. Sudha, N. Nalini, Eur. J. Cancer Prev. 23 (2014) 361 (https://www.doi.org/10.1097/CEJ.0b013e32836473ac)
P. Su, V. P. Veeraraghavan, S. Krishna Mohan, W. Lu, J. Biochem. Mol. Toxicol. 33 (2019) e22403 (https://doi.org/10.1002/jbt.22403)
H. A. Elhady, H. F. Al-Shareef, Mini-Rev. Med. Chem. 20 (2020) 1929 (https://www.doi.org/10.2174/1389557520666200611093510)
Kurasaka, Y. Ogino, A. Sato, Int. J. Mol. Sci. 22 (2021) 2916 (https://www.doi.org/10.3390/ijms22062916)
J. M. Mayer, H. Van De Waterbeemd, Environ. Health Perspect. 61 (1985) 295 (https://www.doi.org/10.1289/ehp.8561295)
M. R. Naylor, A. M. Ly, M. J. Handford, D. P. Ramos, C. R. Pye, A. Furukawa, V. G. Klein, R. P. Noland, Q. Edmondson, A. C. Turmon, W. M. Hewitt, J. Schwochert, C. E. Townsend, C. N. Kelly, M. J. Blanco, R. S. Lokey, J. Med. Chem. 61 (2018) 11169 (https://www.doi.org/10.1021/acs.jmedchem.8b01259)
O. O. Krasnovskaya, Y. V. Fedorov, V. M. Gerasimov, D. A. Skvortsov, A. A. Moiseeva, A. V. Mironov, E. K. Beloglazkina, N. V. Zyk, A. G. Majouga, Arab. J. Chem. 12 (2019) 835 (https://www.doi.org/10.1016/j.arabjc.2016.04.013).