Adsorption analysis of PFOA on activated carbon and ion-exchange resin: A comparative study using four isotherm models Scientific paper
Main Article Content
Abstract
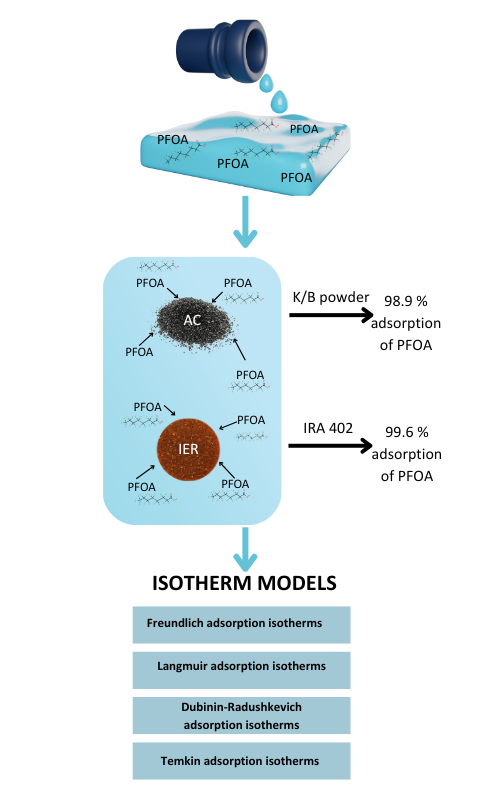
Per- and polyfluoroalkyl substances (PFAS), known as “forever chemicals”, are highly persistent environmental pollutants due to their strong carbon–fluorine bonds. Widely used across industries and consumer products, PFAS have accumulated in the environment, raising concerns about their bioaccumulation, toxicity and mobility. Adsorption, particularly using activated carbon and ion exchange resins, is a suitable technique for PFAS removal from contaminated water. This study evaluates the sorption efficiency of granular and powdered activated carbon and two ion exchange resins to identify the most effective materials for remediation. All tested sorbents showed great performance, however Amberlite IRA 402, and powdered activated carbon K/B were the most efficient. Based on the isotherm models used, it is suggested that physisorption is a dominant process, where the multilayer adsorption on a heterogeneous surface is being favoured.
Downloads
Metrics
Article Details

This work is licensed under a Creative Commons Attribution 4.0 International License.

Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution license 4.0 that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal.
References
Z. Wang, A. M. Buser, I. T. Cousins, S. Demattio, W. Drost, O. Johansson, K. Ohno, G. Patlewicz, A. M. Richard G. W. Walker, G. S. White, E. Leinala, Environ. Sci. Technol. 55 (2021) 15575 (https://doi.org/10.1021/acs.est.1c06896)
L. G. T. Gaines, Am. J. Ind. Med. 66 (2023) 353 (https://doi.org/10.1002/ajim.23362)
I. J. Neuwald, D. Hübner, H. L. Wiegand, V. Valkov, U. Borchers, K. Nödler, M. Scheurer, S. E. Hale, H. P. H. Arp, D. Zahn, Environ. Sci. Technol. 56 (2022) 6380 (https://doi.org/10.1021/acs.est.1c07949)
ECHA, https://echa.europa.eu/hot-topics/perfluoroalkyl-chemicals-pfas (accssed 15.11.2024)
I. Ali, M. Asim, T.A. Khan, J. Environ. Manage. 113 (2012) 170 (https://doi.org/10.1016/j.jenvman.2012.08.028)
N. Bolan, B. Sarkar, Y. Yan, Q. Li, H. Wijesekara, K. Kannan, D.C.W. Tsang, M. Schauerte, J. Bosch, H. Noll, Y. S. Ok, K. Scheckel, J. Kumpiene, K. Gobindlal, M. Kah, J. Sperry, M. B. Kirkham, H. Wang, Y. F. Tsang, D. Hou, J. Rinklebe, J. Hazard. Mater. 401 (2021) 123892 (https://doi.org/10.1016/j.jhazmat.2020.123892)
R. Mahinroosta, L. Senevirathna, J. Environ. Manage. 255 (2020) 109896 (https://doi.org/10.1016/j.jenvman.2019.109896)
F. Dixit, R. Dutta, B. Barbeau, P. Berube, M. Mohseni, Chemosphere 272 (2021) 129777 (https://doi.org/10.1016/j.chemosphere.2021.129777)
E. Barth, J. McKernan, D. Bless, K. Dasu, J. Environ. Manage. 296 (2021) 113069 (https://doi.org/10.1016/j.jenvman.2021.113069)
V. Beškoski, M. Lješević, B. Jiménez, J. Muñoz-Arnanz, P. Colomer-Vidal, H. Inui, T. Nakano, in Soil Remediation Science and Technology. The Handbook of Environmental Chemistry, Vol 130, J. J. Ortega-Calvo, F. Coulon, Eds., Springer Nature, Cham, 2024, p.332 (https://doi.org/10.1007/698_2023_1070)
E. E. Jasper, V. O. Ajibola, J.C. Onwuka, Appl. Water. Sci. 10 (2020) 132 (https://doi.org/10.1007/s13201-020-01218-y)
A. Nastasović, B. Marković, Lj. Suručić, A. Onjia, Metals 12 (2022) 814 (https://doi.org/10.3390/met12050814)
M. Shafiq, A. A. Alazba, M. T. Amin, Sustainability 13 (2021) 3785 (https://doi.org/10.3390/su13073785).